Cell growth, division and differentiation
The main interest of our group is to understand how cells coordinate cell growth and division with cell differentiation. To study this biological problem we use two model organisms, the fission yeast Schizosaccharomyces pombe, a unicellular organism widely used in cell cycle research, and the mouse, where we analyse the physio-pathological consequences of knocking down cell cycle regulator genes in animal cells.
In fission yeast, we have recently described that the Greatwall-Endosulfine (Ppk18-Igo1, in fission yeast) pathway couples the nutritional environment to the cell cycle machinery by regulating PP2A/B55 activity. In the presence of nutrients, Greatwall (Ppk18) protein kinase is inhibited by TORC1 and PP2A/B55 is active. High levels of PP2A/B55 prevent the activation of mitotic Cdk1/Cyclin B and cells increase in size in G2 before undergoing mitosis. When nutrients are limiting, TORC1 activity declines and activation of Greatwall (Ppk18) leads to phosphorylation of Endosulfine (Igo1) and inhibition of PP2A/B55, which in turn allows full activation of Cdk1/Cyclin B and entry into mitosis with a smaller cell size.

The Greatwall-Endosulfine-PP2A/B55 pathway regulates cell size by coupling cell growth (TORC1 activity) to the cell cycle machinery (Cdk1/CyclinB)
In mice we are studying the function of the Anaphase Promoting Complex or Cyclosome (APC/C) and its cofactor Cdh1, an E3 ubiquitin ligase that targets many substrates for proteosomal degradation during G1. Our results indicate that Cdh1-deficient cells accumulate DNA breaks and show slow replication fork progression and increased origin activation, consistent with replication stress. We are currently investigating the role of APC/C-Cdh1 in preventing replicative stress and premature ageing.
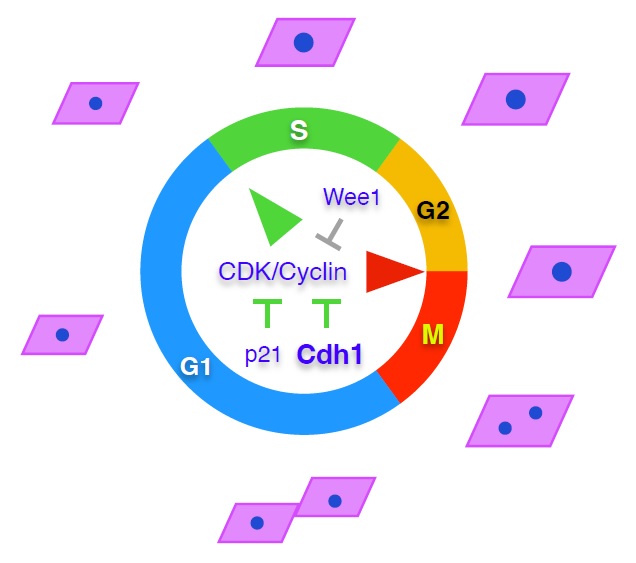
APC/C-Cdh1 prevents premature entry into S-phase, genomic instability and tumour development

Group members
| Sergio Moreno | Research Professor (CSIC) |
|---|---|
| Béla Novák | Professor of Systems Biology, Oxford University |
| Belén Suárez | USAL Senior Lecturer |
| Livia Pérez-Hidalgo | Postdoctoral |
| Javier Encinar del Dedo | Postdoctoral |
| Celia Gálvez-Merchán | PhD Student |
| Rafael López-San Segundo | PhD Student |
| Ana Elisa Rozalén de la Cruz | Technician |
Contact
| Sergio Moreno |
smo@usal.es 923294916 Laboratory 2.6 |
|---|
Recent publications
| Vázquez-Bolado A, López-San Segundo R, García-Blanco N, Rozalén AE, González-Álvarez D, Suárez MB, Pérez-Hidalgo L and Moreno S (2022)
The Greatwall-Endosulfine switch accelerates autophagic flux during the cell divisions leading to G1 arrest and entry into quiescence in fission yeast. International Journal of Molecular Sciences 24: 148 |
| Cuadrado M, Garzón J, Moreno S* and García-Higuera I* (2022) * Co-corresponding authors
Efficient terminal erythroid differentiation requires the APC/C cofactor Cdh1 to limit replicative stress in erythroblasts. Scientific Reports 12: 10489 |
| García-Blanco N and Moreno S (2019)
Down-regulation of Cdk1 activity in G1 coordinates the G1/S gene expression programme with genome replication. Current Genetics 65: 685-690 |
| Rubio A, García-Blanco N, Vázquez-Bolado A, Suárez MB and Moreno S (2018)
Nutritional cell cycle reprogramming reveals that inhibition of Cdk1is required for proper MBF-dependent transcription. Journal of Cell Science 131, jcs218743 |
| Garzón J, Rodríguez R, Kong Z, Chabes A, Rodríguez-Acebes S, Méndez J, Moreno, S*. and García-Higuera, I* (2017) * Co-corresponding authors
Shortage of dNTPs underlies altered replication dynamics and DNA breakage in the absence of the APC/C cofactor Cdh1. Oncogene 36: 5808-5818 |
| Pérez-Hidalgo L and Moreno S (2016)
Nutrients control cell size. Cell Cycle 8: 1-2 |
| Chica N, Rozalén AE, Pérez-Hidalgo L, Rubio A, Novak B and Moreno S (2016)
Nutritional control of cell size by the greatwall-endosulfine-PP2A·B55 pathway. Current Biology 26: 319-330 |
Research grants
| Ministry of Science and Innovation PID2020-115929RB-I00 |
| Junta de Castilla y León CSI259P20 |
| Junta de Castilla y León CL-EI-2021-08 |







